Difference between Chemical and Nuclear Reaction
Key difference: Chemical reaction is a process in which atoms of an element rearrange themselves to form a new substance. Nuclear reaction is a process in which the structure of the nucleus of an atom is changed with the release of energy.
The main difference between nuclear reaction and chemical reaction can be understood on the basis of ‘how the reaction takes place in the atom’. Nuclear Reaction takes place in the atom’s nucleus; whereas the electrons in the atom are responsible for Chemical Reactions.
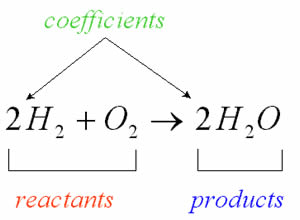 Chemical reaction is a process of converting an element into a whole new element. The element at the beginning of the reaction is known as reactant and the new element formed after the reaction is known as product. When one or more reactants are converted into products, they undergo many modifications and energy changes. The chemical reactions involve the loss, gain and sharing of electrons. This transfer of electrons among the elements are known as chemical reaction. It can also be described using chemical equations.
Chemical reaction is a process of converting an element into a whole new element. The element at the beginning of the reaction is known as reactant and the new element formed after the reaction is known as product. When one or more reactants are converted into products, they undergo many modifications and energy changes. The chemical reactions involve the loss, gain and sharing of electrons. This transfer of electrons among the elements are known as chemical reaction. It can also be described using chemical equations.
There are various factors controlling the equations such as catalysts, temperature, solvent effects, etc. Through the study of thermodynamics, the reactions can be controlled. Rate of reaction simply determines the speed of a reaction. It is a parameter which studies, how slow or fast a reaction is. Taking an example,
Consider the following reaction where reactants A and B are going into products C and D.
a A + b B → c C + d D
The rate for the reaction can be given in terms of either of two reactants or products.
Rate = -1/a × d[A]/dt= -1/b × d[B]/dt = 1/c × d[C]/dt = 1/d × d[D]/dt
Here, a, b, c and d are the coefficients of the reactants and products. For the reactants, there is a minus sign, because they are depleting
as the reaction proceeds. However, as the products are increasing, they are given positive signs.
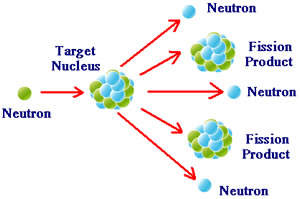
Nuclear reactions involve the decomposition of the nucleus, as it decomposes the atom; it forms a new element with the loss of protons or neutrons. The reaction takes place inside the nucleus. There is a very high energy change in a nuclear reaction. It can be categorized into, nuclear fusion and nuclear fission. In fusion, two types of nuclei are combined to release energy. In fission, a large unstable nucleus is split into two smaller stable nuclei and energy is released.
Comparison between Chemical and Nuclear Reaction:
|
|
Chemical Reaction |
Nuclear Reaction |
|
Definition |
Elements rearrange themselves to form a new element. |
Structure of nucleus is changed with release of energy. |
|
Reaction |
Electrons are responsible for the reaction. |
It takes place in the nucleus of atom. |
|
Position |
It takes place outside the nucleus. |
It takes place inside the nucleus. |
|
Nature |
Involves loss, gain and sharing of electrons. |
Involves the decomposition of nucleus. |
|
Energy |
Low energy change. |
High energy change. |
|
Isotopes |
They react the same |
They react differently. |
|
Chemical combinations |
It is depended. |
It does not depend. |
|
Mass |
Reactants and products have equal mass. |
There is mass change. |
Image Courtesy: tutorvista.com, zube.brinkster.net









Comments
When you are discussing chemical reactions, you keep using the term "element" when you mean "molecule". In a chemical reaction, elements in one or more molecule rearrange themselves into one or more different molecules.
Mrs. Stephenson
Sun, 05/10/2015 - 20:24
Add new comment